Manual review, algorithmic detection, and the importance of validation
Previously, we explored What Is Atrial Fibrillation Burden? explaining the concept and its clinical significance. If you haven’t read that piece yet, it’s worth checking out for background. As previously discussed, AF burden quantifies the proportion of time a patient spends in atrial fibrillation and provides clinically meaningful information beyond a binary AF diagnosis, including insights into stroke risk, heart failure progression, and treatment response.
The next critical question is how this parameter is calculated in practice. In this article, we will focus on how to calculate AF Burden. We will compare the traditional manual review of ECG recordings with modern algorithmic detection, and introduce Cardiomatics’ validated approach for accurate and reliable AF burden assessment.
Why measuring AF burden matters
AF is rarely a static condition. Patients may have brief, intermittent episodes or prolonged periods of arrhythmia that go unnoticed. Understanding the extent of AF over time is crucial: it helps clinicians assess risk, monitor disease progression, evaluate treatment effectiveness, and guide decisions on anticoagulation or rhythm-control therapy.
Quantifying AF as a single number – its burden – translates complex ECG data into an actionable metric. Unlike a simple yes/no diagnosis, AF burden reflects the real-world load of arrhythmia, providing insights that can influence both clinical outcomes and patient quality of life.
It is important to note that the clinical meaning of AF burden depends on the clinical context. The same AF burden value may have different implications in different patients. For example, in patients with a prior stroke or TIA, even a relatively low AF burden may be clinically relevant and support anticoagulation decisions. In contrast, after catheter ablation, AF burden is often used to track treatment effectiveness and rhythm control over time.
Similarly, AF burden should be interpreted together with clinical risk factors and established scores, rather than as a standalone parameter. Its value lies in supporting clinical decisions within the broader clinical context.
How to calculate AF burden?
Calculating AF burden is fundamentally a process of accurate rhythm classification over time. The calculation itself is simple, but the challenge lies in the detection.
First, the total analyzable ECG time must be established. Then all the segments that are affected by excessive noise or signal loss should be excluded. The rest of the recording is checked to see if AF is present or not.
AF burden is calculated by summing the duration of all AF-classified segments and expressing this value relative to the total analyzable monitoring time. Even minor inaccuracies in rhythm classification can meaningfully alter the final burden estimate, particularly in long recordings.
For this reason, the reliability of AF burden depends primarily on how AF episodes are detected, rather than on the mathematical formula used to calculate the burden itself.
Manual ECG review: strengths and limitations
Manual ECG review has generally been considered a clinical reference standard for AF detection. Trained reviewers visually inspect the recordings and identify AF using established electrocardiographic criteria.
The approach entails several obvious advantages. Human interpretation allows for contextual judgment, differentiation of AF from other irregular rhythms, and informed decisions in the event of suboptimal signal quality. However, manual review is resource-intensive and scales poorly. Multi-day or continuous recordings require substantial time and concentration, and inter-observer variability remains an inherent limitation. With increasing duration of monitoring, feasibility decreases with manual analysis, particularly in routine clinical practice.
AF detection using algorithms
The development of the Algorithmic AF detection was driven by the growing volume of ECG data. Automated algorithms evaluate the level of rhythm irregularity, the presence of atrial activity, and signal quality to identify AF episodes efficiently and consistently.
On the one hand, algorithmic detection offers important advantages for AF burden estimation:
- rapid analysis of long recordings,
- reproducible results,
- standardized burden calculation.
Although automated algorithms provide efficient and scalable AF burden assessment, their performance can be influenced by signal complexity, including noise, or frequent ectopy. For this reason, validation within clinically representative datasets remains an important step to ensure robust interpretation.
Manual vs algorithmic AF burden assessment
Manual and algorithmic approaches should not be positioned as competing methods, but as complementary tools. Manual review provides interpretive depth, while algorithmic detection offers scalability and consistency.
In modern workflows, algorithmic detection is increasingly used as the foundation for AF burden estimation, with expert review reserved for ambiguous or clinically critical segments.
The key determinant of clinical usefulness is not whether detection is manual or automated, but whether it is accurate, reproducible, and validated.
The role of validation in AF burden estimation
Accurate AF burden estimation depends far more on validated detection performance than on the burden formula itself. Validation defines whether an AF burden value can be trusted in clinical decision-making or should be interpreted with caution.
From a clinical perspective, validation answers three critical questions:
- Does the system correctly identify AF across diverse ECG conditions?
AF detection must perform reliably in the presence of noise, baseline wander, motion artefacts, ectopy, and varying heart rates. Algorithms that are not validated on heterogeneous, real-world datasets may systematically overestimate or underestimate AF burden. - Can AF be reliably distinguished from look-alike rhythms?
Atrial flutter, frequent premature atrial contractions, sinus arrhythmia, and paced rhythms often mimic AF in RR-interval–based analyses. Without validation against expert-labelled reference data, misclassification of these rhythms can significantly distort AF burden values. - Is performance stable across long-term recordings?
AF burden is particularly sensitive to cumulative error. Even small classification inaccuracies, when applied over days or weeks of monitoring, can lead to clinically meaningful deviations in burden estimates.
Cardiomatics validated approach to AF burden calculation
Cardiomatics’ approach to AF burden calculation is designed to reflect how cardiologists actually interpret long-term ECG recordings: by combining algorithmic efficiency with ECG-experts manual review.
At the core of the Cardiomatics system is a validated AF detection algorithm that analyzes the ECG signal on a beat-to-beat basis, integrating:
- R–R interval variability,
- atrial activity characteristics,
- signal quality assessment,
- and context-aware rhythm classification.
Before AF burden is calculated, the system determines the total good-quality analyzable signal duration, explicitly excluding segments affected by noise, artefacts, or signal loss. This step is critical, as AF burden is always expressed relative to reliable ECG data, not raw recording time.
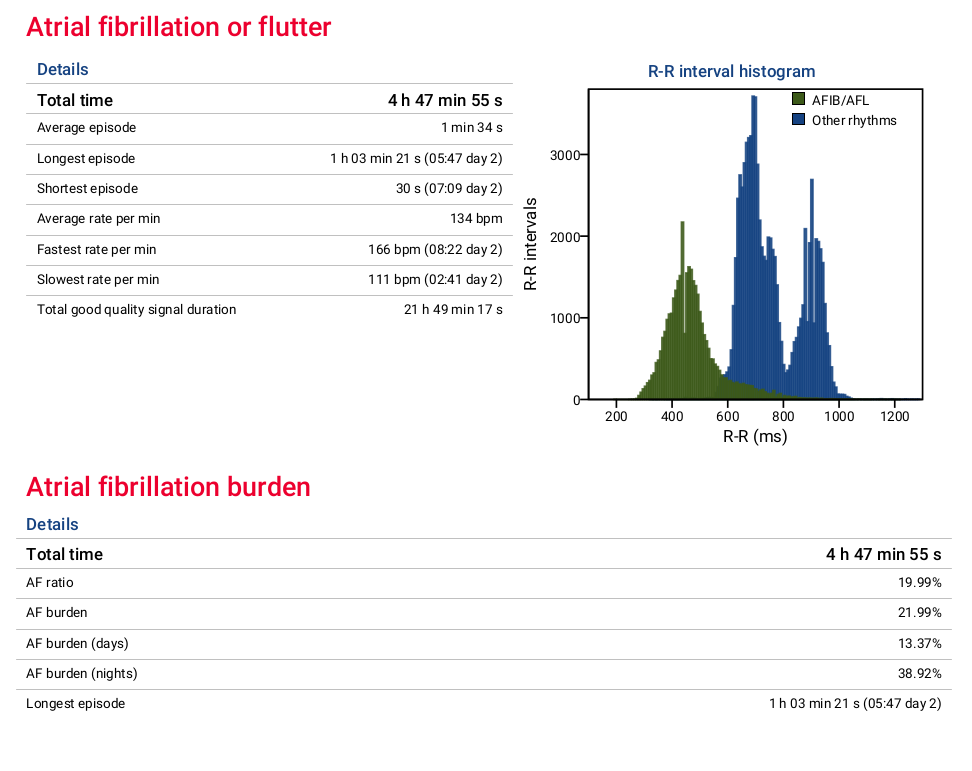
The Cardiomatics clinical report — as illustrated in the example above — presents AF burden in a clear, interpretable, and clinically relevant format:
Key elements include:
- Total AF/AFL time across the recording,
- AF burden (%) relative to good-quality signal duration,
- Daytime vs nighttime AF burden, enabling circadian pattern analysis,
- Episode statistics, including longest, shortest, and average AF episodes,
- Heart rate characteristics during AF, providing additional physiological context.
The inclusion of R-R interval histograms further enhances interpretability by visually separating AF/AFL rhythm patterns from other rhythms, supporting both algorithmic classification and clinician confidence.
Importantly, Cardiomatics’ workflow does not treat automation as a black box. Algorithmic detection forms the basis, with expert review focused on diagnostically challenging or clinically critical segments. This hybrid approach ensures scalability without sacrificing diagnostic reliability. By combining validated algorithms, strict signal-quality control, and transparent reporting, Cardiomatics delivers AF burden estimates that are not only numerically accurate, but also clinically meaningful and actionable.
Conclusions
AF burden gives a clear, meaningful picture of a patient’s atrial fibrillation, showing the true arrhythmic load rather than a simple yes/no answer. Its reliability comes from validated detection methods that combine algorithmic precision with expert review. When measured carefully, AF burden helps clinicians assess risk, guide treatment decisions, and provide personalized care. By integrating rigorous signal-quality control, transparent reporting, and hybrid review, AF burden becomes not just a number, but a trusted tool for actionable insights in everyday practice.

